Flow Cytometry solutions for evaluating CAR T-cell expression
What are CAR T-cells?
CAR T-cells, also known as chimeric antigen receptor T cells, are a type of immunotherapy used in the treatment of certain types of cancer. They play a crucial role in the body's immune system by recognizing and eliminating abnormal cells, including cancer cells.
CAR T-cell therapy genetically alters a patient's T cells with a chimeric antigen receptor (CAR), a synthetic receptor, fusing antibody precision with the T cell's immune-triggering capacity. This CAR targets and attaches to cancer cell surface proteins, activating the T-cell for a robust immune assault and eventual cancer cell eradication.
CAR T-cell immunotherapy created new hope in the fight against cancer. As of September 2023, there are six CAR T-cell therapies approved by the FDA. While these therapies target B-cell cancers and multiple myeloma, numerous ongoing clinical trials are investigating the efficacy of CAR T–cell therapies against solid tumors1.
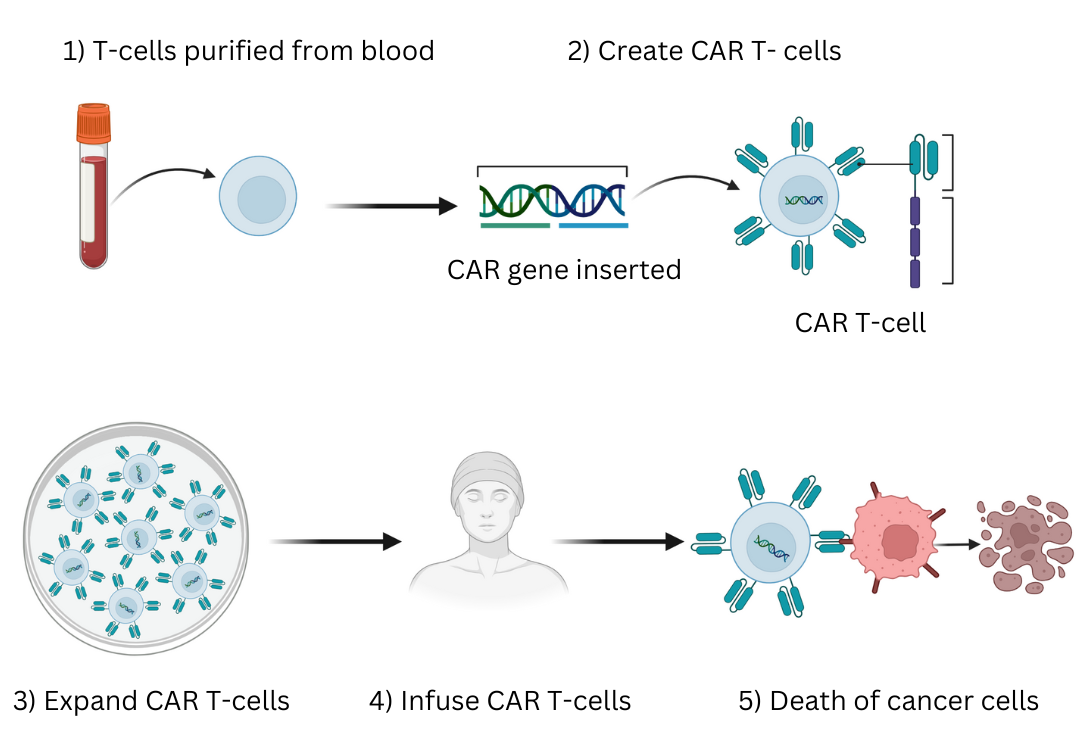
Generating CAR T-cells involves several steps. T cells are first isolated from a donor, activated, and transduced to insert the CAR gene. The cells are then expanded, and a number of tests are done to confirm characteristics of the transduced cells, including whether the T-cells are expressing the CAR.
How is CAR T-cell expression evaluated?
Evaluation of CAR protein expression is critical, since without proper expression, the CAR T-cell cannot function as intended, to eliminate the targeted cancer cell. CAR protein expression is confirmed by a number of methods, including flow cytometry, using CAR target antigens conjugated to a fluorescent tag such as Phycoerythrin (PE) or Allophycocyanin (APC). This direct detection approach is a preferred choice because of low background and high specificity for the CAR. However, indirect methods using biotinylated or unconjugated antigens in combination with fluorescently labeled antibodies provide additional options for detection.
OriGene solutions for accelerating CAR T- cell expression verification
Choose your Flow Cytometry detection option:
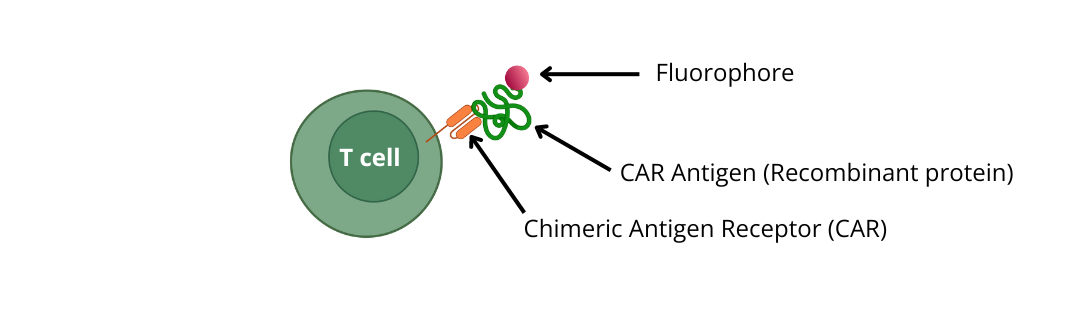
1) Use recombinant proteins (CAR antigens) conjugated to a fluorophore
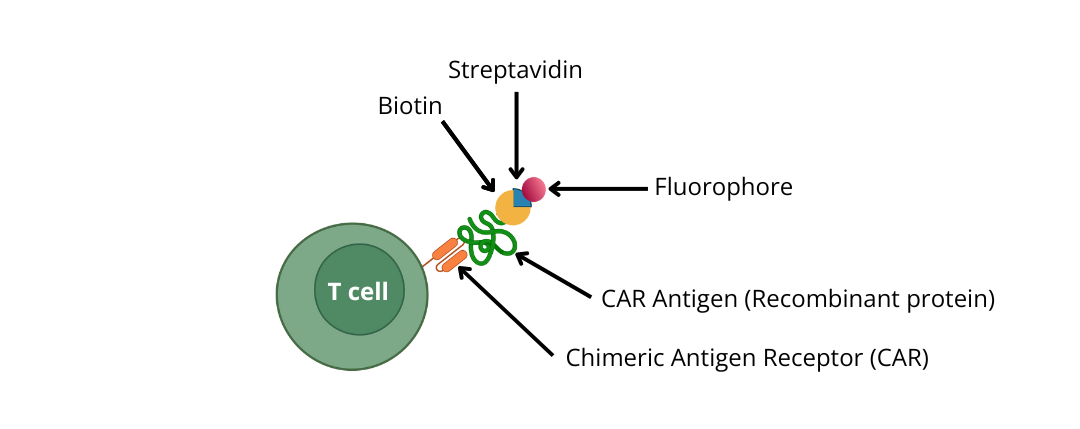
2) Use biotinylated recombinant proteins (CAR antigens) and detect with Streptavidin conjugated to a fluorophore
3) Use epitope -tagged recombinant proteins (CAR antigens) and detect with an anti-tag antibody conjugated to a fluorophore
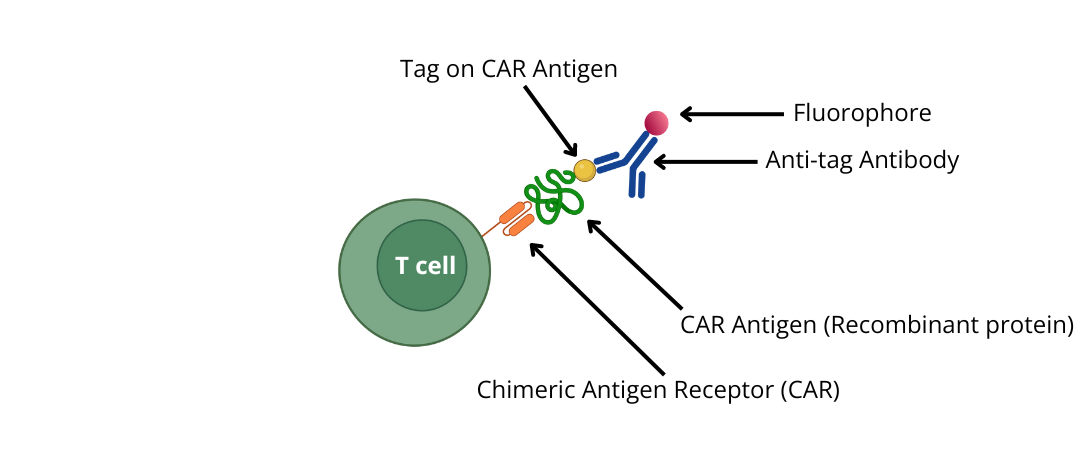
Search for additional CAR Targets and corresponding antibodies.
Detection of PE conjugated BCMA recombinant protein by flow cytometry.
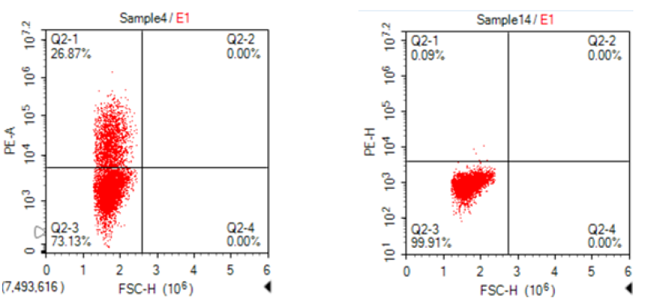
A: BCMA CAR – transfected CHO cells B: Mock-transfected CHO cells
Transfected CHO cells were stained with PE conjugated BCMA-Fc protein () at 4.0 µg/mL for 1 hr at RT.
Learn about our custom solutions to accelerate your CAR T research.
References:
1. Mitra A, Barua A, Huang L, Ganguly S, Feng Q, He B. From bench to bedside: the history and progress of CAR T cell therapy. Front Immunol. 2023 May 15;14:1188049. doi: 10.3389/fimmu.2023.1188049. PMID: 37256141; PMCID: PMC10225594.
Diagrams generated with Biorender
