CytoSections™
What are CytoSections?
CytoSections are FFPE sections of transiently transfected cDNA-specific gene targets expressed in human HEK293T cells, providing dependable and renewable positive control solutions to the IHC, ICC, and ISH users.
When can CytoSections be used?
-
Positive Control for IHC/ICC/ISH Workflow:
Eliminate positive control hunting - CytoSections integrate into your existing workflow (click here to learn more)
-
Validate Antibodies Faster:
Screen large pools for specific antibodies, including for those against rare mutations, without wasting tissues, time, or money (click here to learn more)
-
Multicenter Reference:
Ideal reference for consistent results across labs
Key Features and Benefits:
- Study Rare Gene Mutations:
- Access hard-to-find gene mutations with custom CytoSections
- Save Precious Tissue, Time, and Money
- Start experiments right away knowing that target expression for each lot is verified
- FFPE sections are comparable to your samples and easily incorporated into existing workflows
- Do not waste precious tissue for assay optimization and costly method development
- Once your assay is developed, cost effectively obtain as many slides as you need
- Increase Your Confidence in Generating Reproducible Date
- Transient transfection ensures consistent gene expression, unlike stable cell lines where expression can change over time, creating lot to lot variability
- No section-to-section expression variability
How are CytoSections made for your specific gene target?
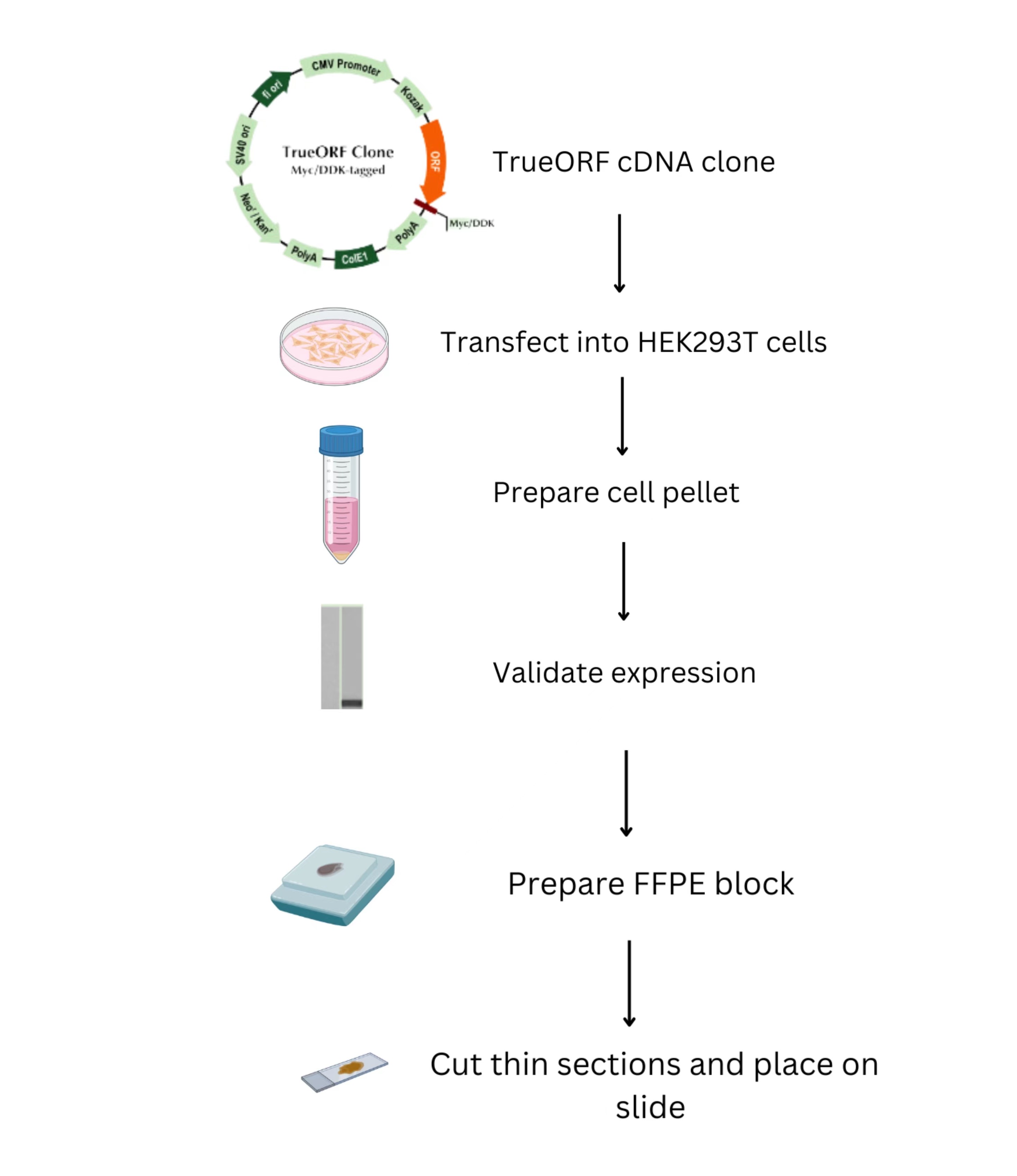
TrueORF cDNA clones containing the gene of interest (GOI) are transiently transfected into mammalian cells. The transfected cells are spun down into a cell pellet, which is tested to validate the expression of the GOI before creating the FFPE block. Subsequently, the block is cut into thin slices and placed on a slide.
CytoSection control slides maintain the cellular localization patterns of nuclear and cytoplasmic/membrane-associated proteins, closely mirroring their distribution in tissue samples.
Nuclear localization of BRG1
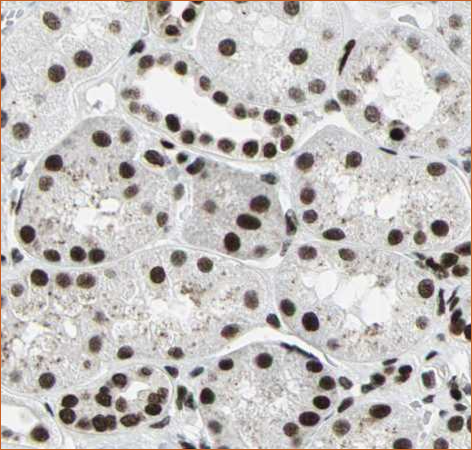
BRG1 antibody of kidney tissue. Image source: Protein atlas
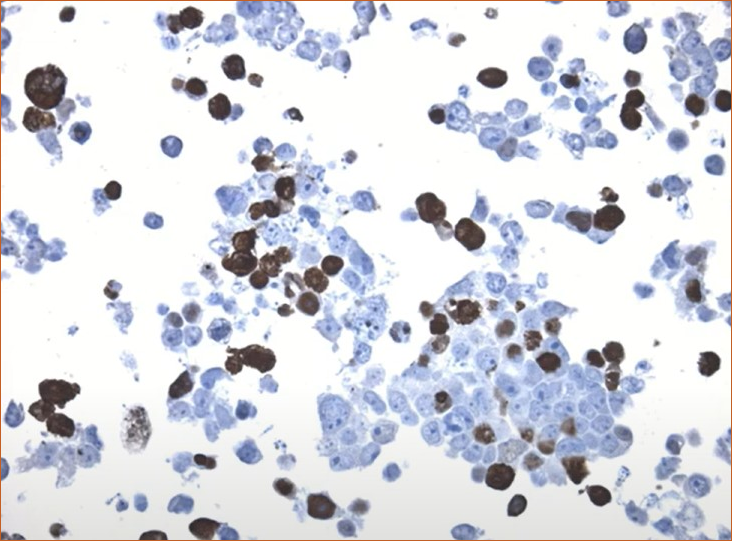
DDK-tag detection of BRG1 in CytoSection ()
Cytoplasmic localization of DCC
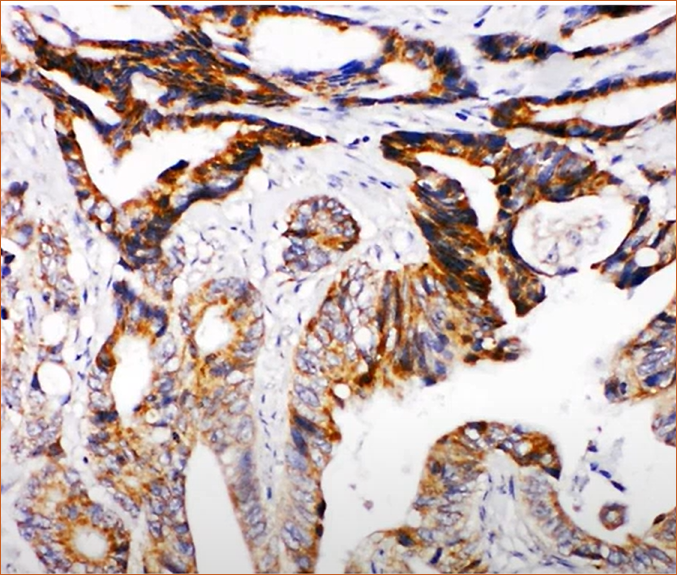
DCC antibody of colon tissue. Image source: Protein atlas
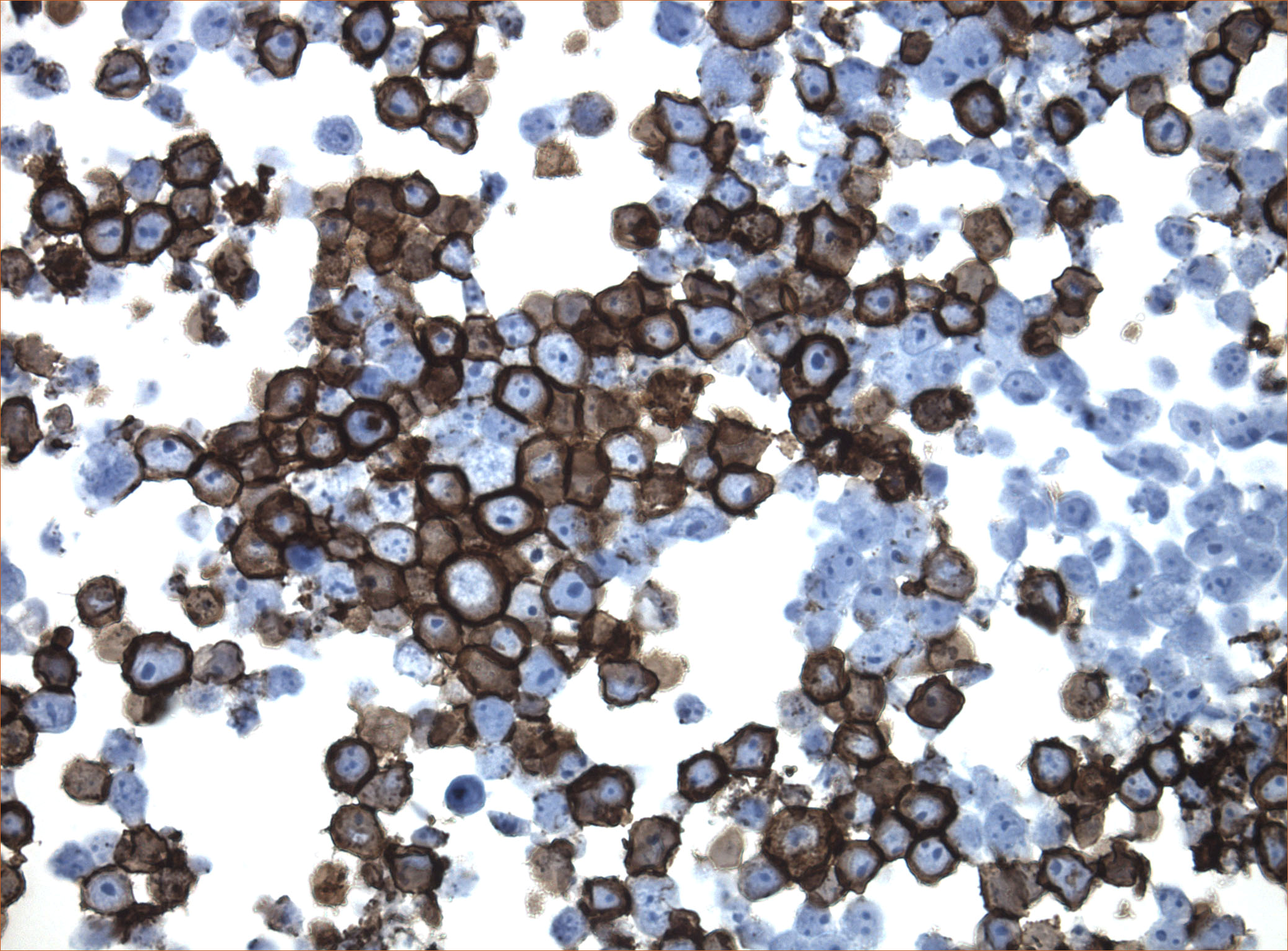
DDK-tag detection of DCC in CytoSection ()
A major challenge with tissue slides is reproducibility due to the inherent heterogeneity of tissue samples. CytoSection control slides, derived from transiently transfected cells, ensure consistent gene expression and superior lot-to-lot reproducibility, making them a reliable alternative for researchers.
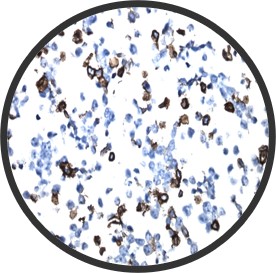
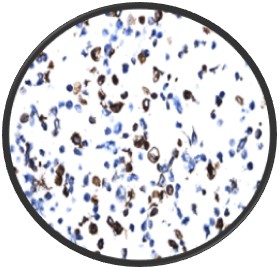
Two different lots of ENPP1 CytoSections () showed reproducible gene expression
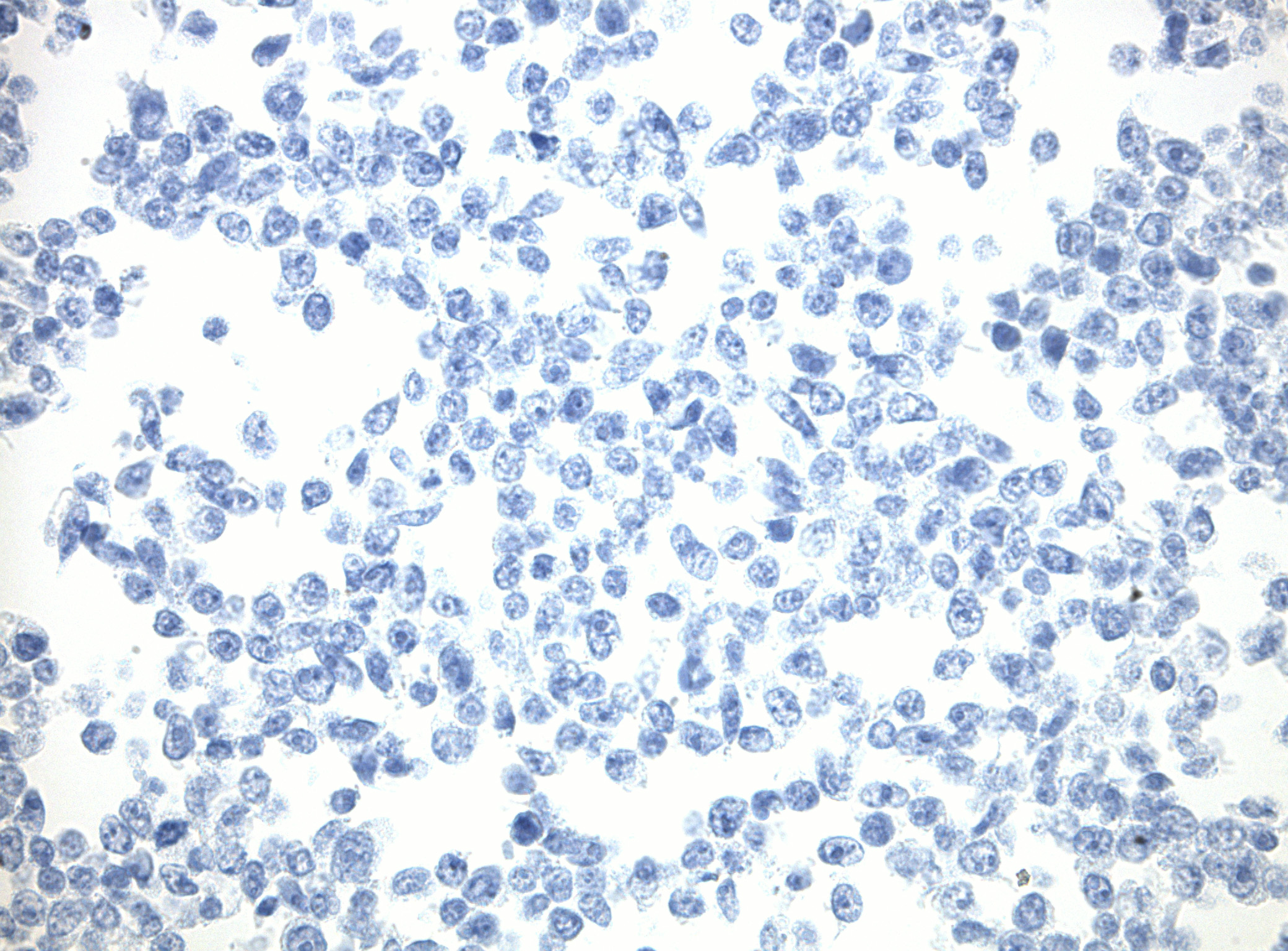
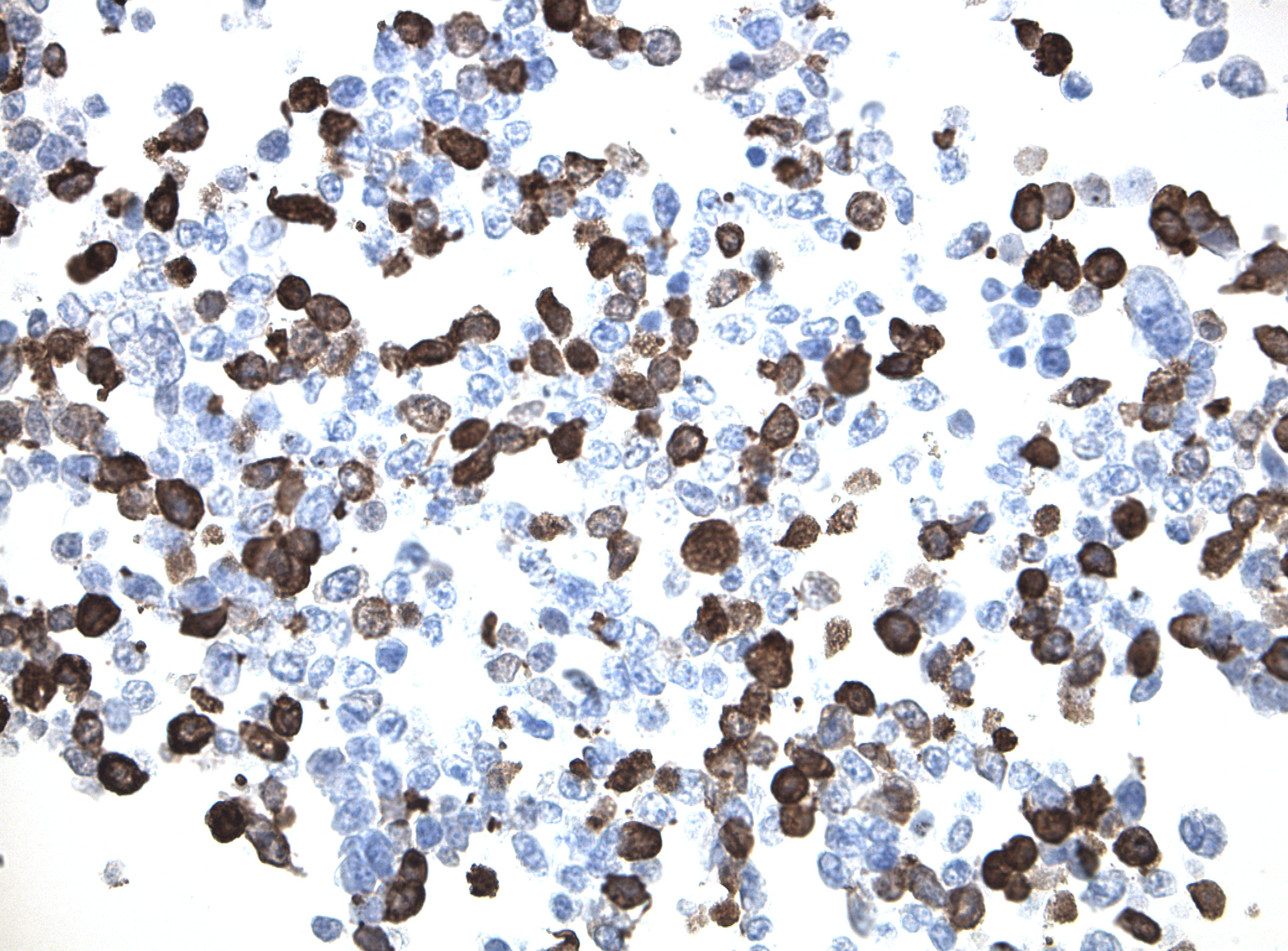
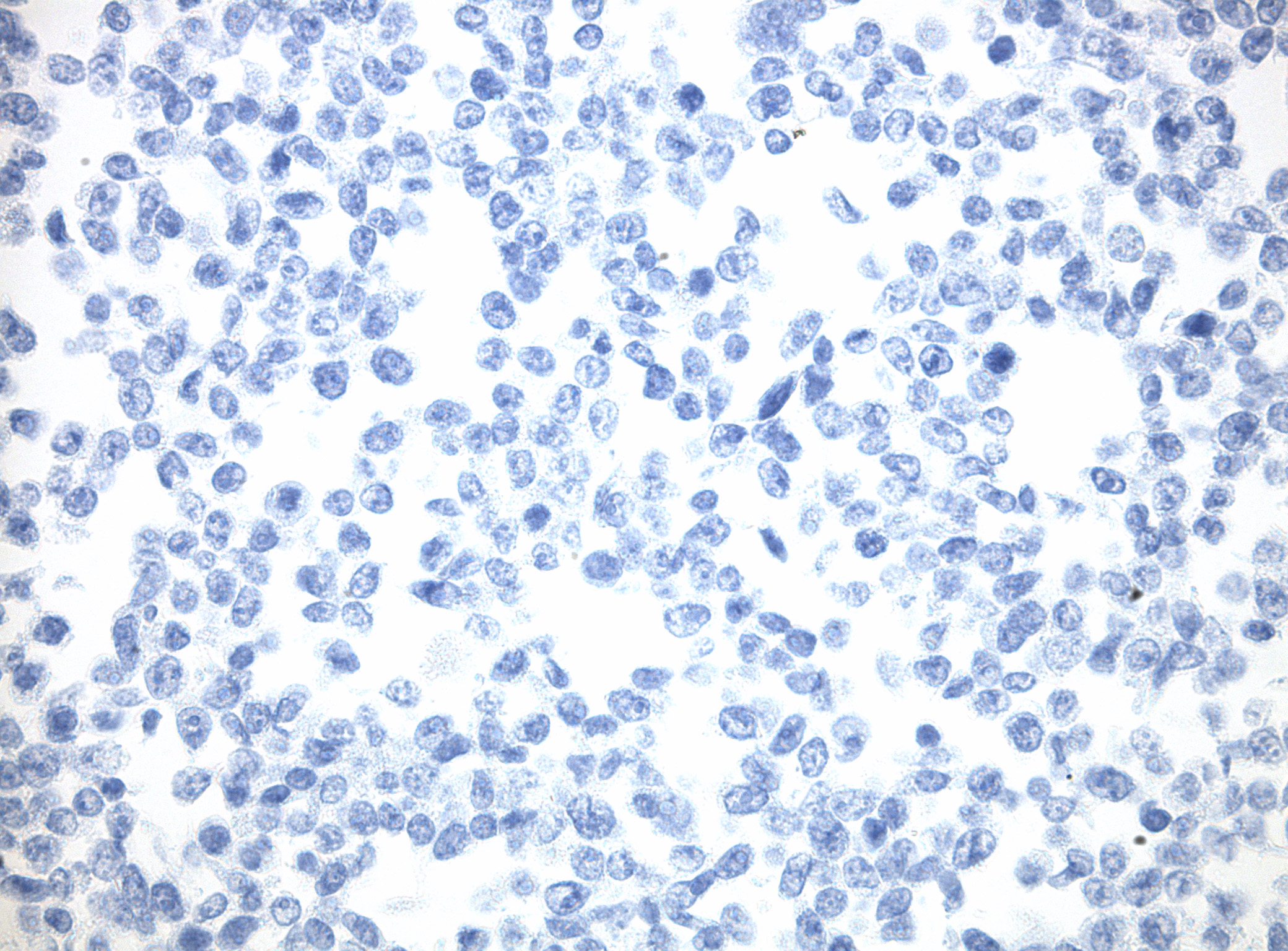
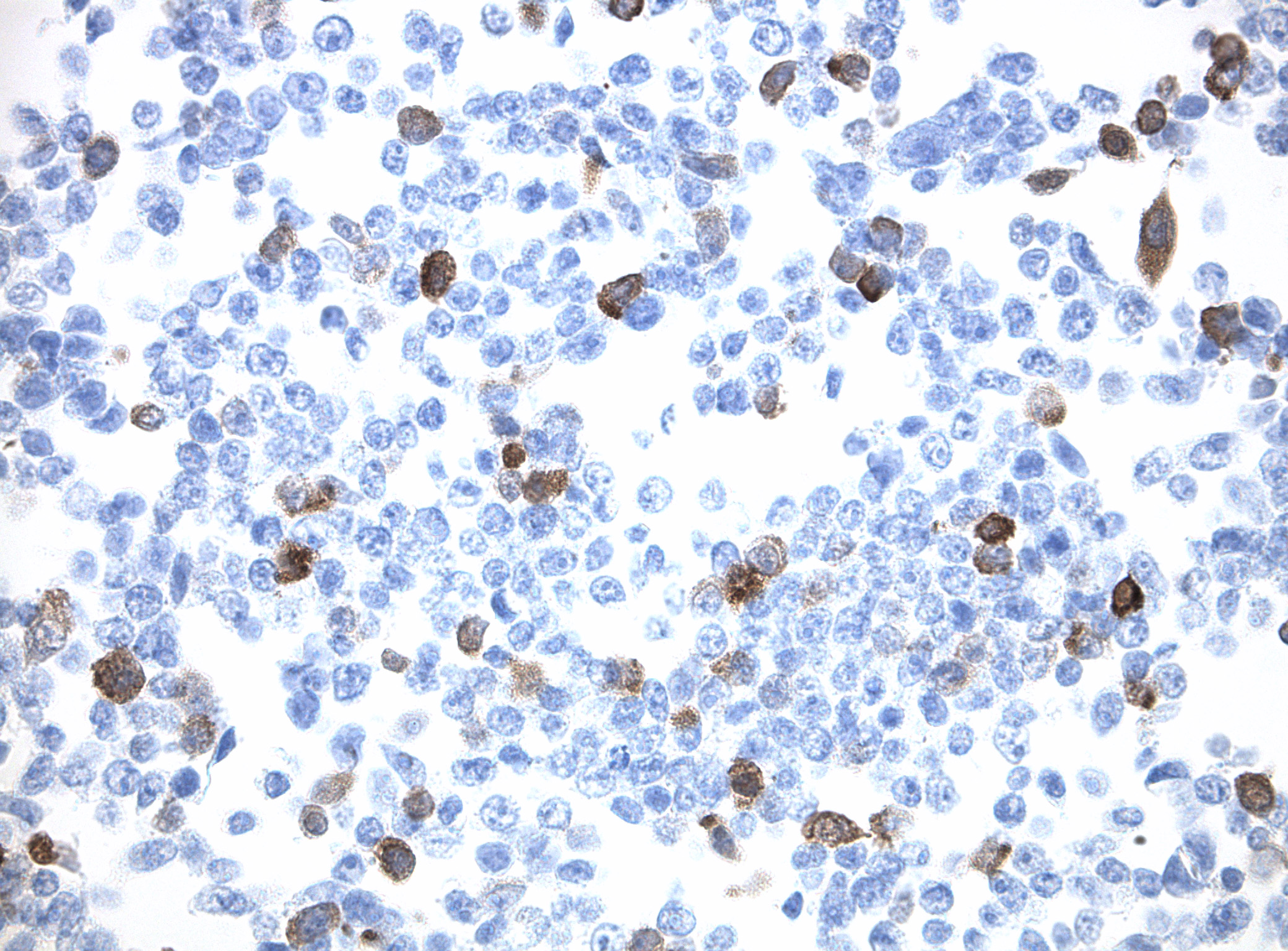
IDH1 (R132H) Mutant-Specific Antibody Screening, Validation, and Specificity
CytoSections enables identifying the right antibody for your research. See how CytoSections, featuring WT or mutant IDH1 (R132H) protein, helped screening and validation of specific antibody against the mutant IDH1 (R132H).
|
|
|
|
|
|
CytoSections |
IDH1 (WT) |
IDH1 (R132H) |
IDH1 (WT) |
IDH1 (R132H) |
|
IDH1 (R132H) Antibody |
|
|
||
|
Reactivity |
None |
Strong |
None |
Mild |
CytoSections as In-situ Hybridization (ISH) Control
In CytoSections, RNA expression is preserved alongside protein expression, making them ideal as positive and negative controls for in situ hybridization (ISH) applications. The case study below demonstrates positive staining on ENPP1 CytoSections () using an ENPP1 RNA probe, while no staining is observed with the same probe on ENPP2 CytoSections (), confirming specificity.
ENPP1 CytoSections
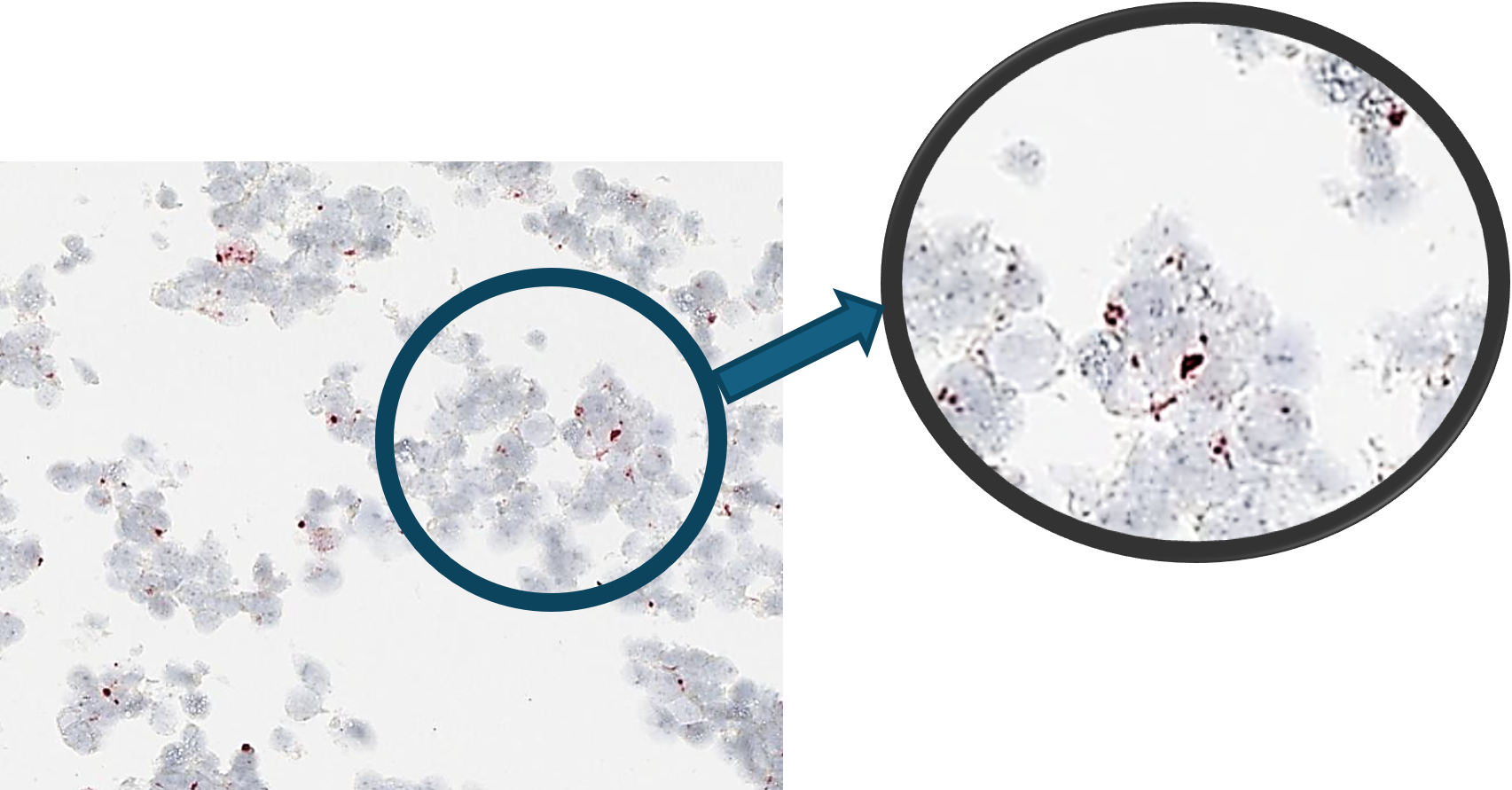
Positive Stained: ISH-based ENPP1 RNA probe
ENPP1 CytoSections
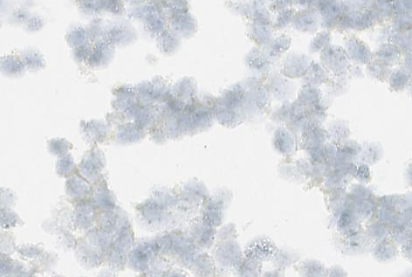
Negative Stained: dapB Control Probe
ENPP2 CytoSections
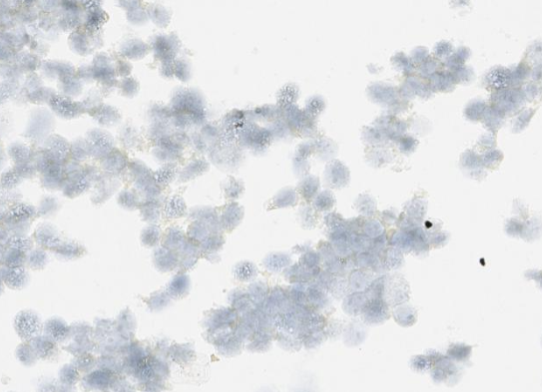
Negative Stained: ISH-based ENPP1 RNA probe
CytoSections as Immunofluorescent (IF) Control
CytoSections can be utilized beyond IHC and ISH applications. They integrate seamlessly into immunofluorescence (IF) workflows, serving as reliable controls for workflow validation and antibody specificity testing in IF experiments. The case study below demonstrates the use of an Alexa Fluor 647-conjugated anti-MAGEA3 antibody to detect MAGEA3 in a MAGEA3 CytoSection ().
MAGEA3 CytoSections
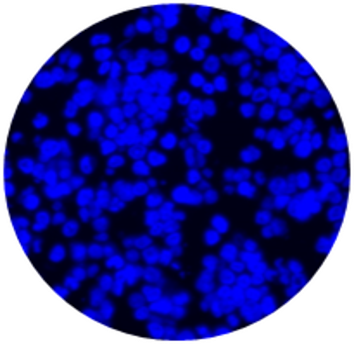
DAPI

Anti-MAGEA3 Antibody
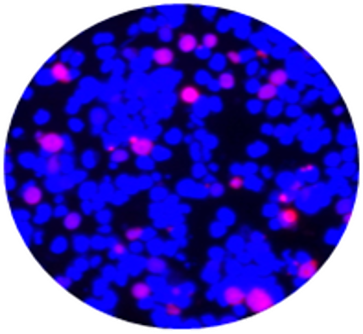
Overlay of DAPI/Anti-MAGEA3 Antibody
CytoSections can serve as effective immunofluorescence (IF) controls for validating antibody specificity. The case study below demonstrates the use of an Alexa Fluor 488-conjugated anti-MAGEA4 antibody to detect MAGEA4 in MAGEA4 CytoSections (, while showing no cross-reactivity in MAGEA3 CytoSections ().
MAGEA4 CytoSections (Positive Signal)

DAPI
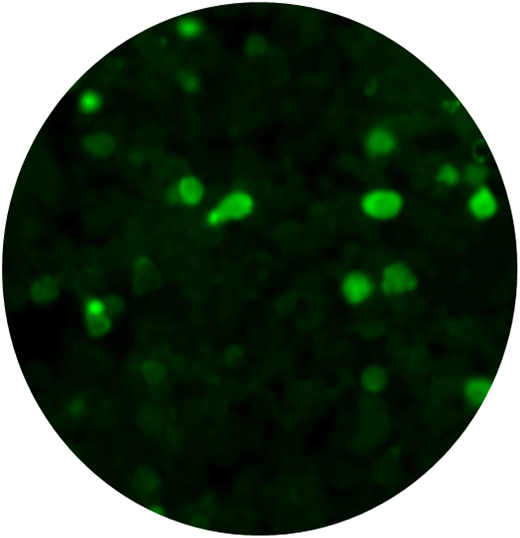
Anti-MAGEA4 Antibody
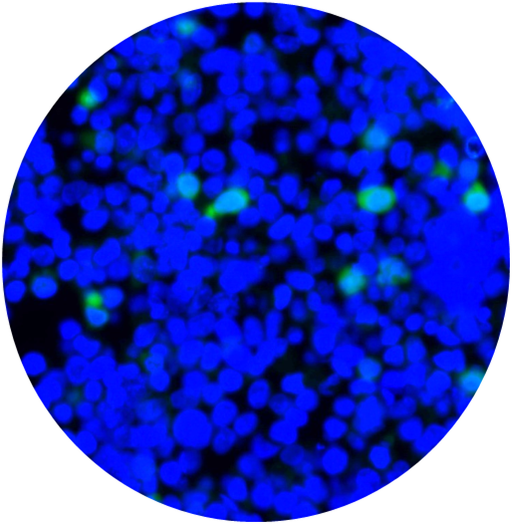
Overlay of DAPI/Anti-MAGEA3 Antibody
MAGEA3 CytoSections (No Signal)
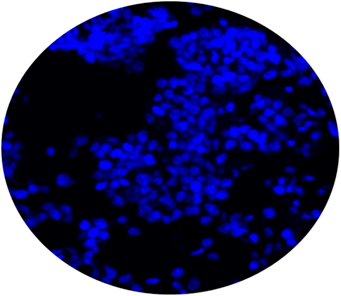
DAPI

Anti-MAGEA4 Antibody
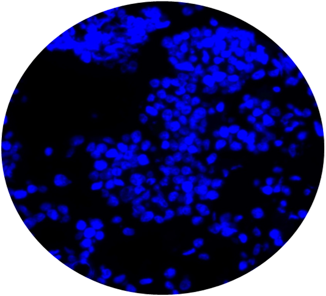
Overlay of DAPI/Anti-MAGEA3 Antibody
CytoSections as IHC Control
Choose from hundreds of CytoSections as dependable controls for your immunohistochemistry (IHC) experiments. Easily integrate them into your assay workflow to achieve consistent, reproducible results. Replace traditional tissue controls with CytoSections for enhanced reliability and standardization in IHC applications.
The case study below demonstrates the use of an anti-MAGEA3 antibody to detect MAGEA3 in a MAGEA3 CytoSection () and negative staining on untransfected HEK293T CytoSection ().
.png)
.png)
CytoSections offer positive controls for a vast library of genes. If you do not see your desired gene of interest, fill out our custom quote form, and our experienced scientists will design CytoSections to fit your needs.
Related Products
| CytoSections™ Resourcess |
