LAP3 (NM_015907) Human Recombinant Protein
CAT#: TP309052
Recombinant protein of human leucine aminopeptidase 3 (LAP3), 20 µg
Frequently bought together (2)
Other products for "LAP3"
Specifications
| Product Data | |
| Species | Human |
| Expression Host | HEK293T |
| Expression cDNA Clone or AA Sequence |
>RC209052 protein sequence
Red=Cloning site Green=Tags(s) MFLLPLPAAGRVVVRRLAVRRFGSRSLSTADMTKGLVLGIYSKEKEDDVPQFTSAGENFDKLLAGKLRET LNISGPPLKAGKTRTFYGLHQDFPSVVLVGLGKKAAGIDEQENWHEGKENIRAAVAAGCRQIQDLELSSV EVDPCGDAQAAAEGAVLGLYEYDDLKQKKKMAVSAKLYGSGDQEAWQKGVLFASGQNLARQLMETPANEM TPTRFAEIIEKNLKSASSKTEVHIRPKSWIEEQAMGSFLSVAKGSDEPPVFLEIHYKGSPNANEPPLVFV GKGITFDSGGISIKASANMDLMRADMGGAATICSAIVSAAKLNLPINIIGLAPLCENMPSGKANKPGDVV RAKNGKTIQVDNTDAEGRLILADALCYAHTFNPKVILNAATLTGAMDVALGSGATGVFTNSSWLWNKLFE ASIETGDRVWRMPLFEHYTRQVVDCQLADVNNIGKYRSAGACTAAAFLKEFVTHPKWAHLDIAGVMTNKD EVPYLRKGMTGRPTRTLIEFLLRFSQDNA TRTRPLEQKLISEEDLAANDILDYKDDDDKV |
| Tag | C-Myc/DDK |
| Predicted MW | 56 kDa |
| Concentration | >0.05 µg/µL as determined by microplate BCA method |
| Purity | > 80% as determined by SDS-PAGE and Coomassie blue staining |
| Buffer | 25 mM Tris-HCl, 100 mM glycine, pH 7.3, 10% glycerol |
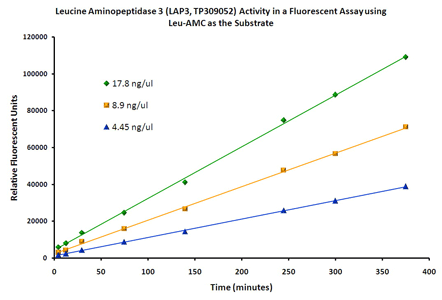
| Bioactivity | LAP3 activity verified in a biochemical assay: Leucine aminopeptidase 3 (LAP3, TP309052) activity was measured in a fluorescent biochemical assay. LAP3 catalyzes the removal of unsubstituted N-terminal amino acids from various peptides and is most active on leucine. LAP3 activity was measured in a 100 µl reaction mixture containing 1 mM L-leucine 7-amido-4-methyl coumarin (Leu-AMC), 50 mM Tris, pH 8.0, 4 mM MgCl2, and 1 mM MnCl2. Cleavage of leucine from the AMC moiety results in a strong increase in fluorescence intensity. Fluorescence was measured over time with an excitation wavelength of 380 nm and an emission wavelength of 460 nm. The activity of the enzyme in this system remained constant over six hours. |
| Preparation | Recombinant protein was captured through anti-DDK affinity column followed by conventional chromatography steps. |
| Note | For testing in cell culture applications, please filter before use. Note that you may experience some loss of protein during the filtration process. |
| Storage | Store at -80°C. |
| Stability | Stable for 12 months from the date of receipt of the product under proper storage and handling conditions. Avoid repeated freeze-thaw cycles. |
| Reference Data | |
| RefSeq | NP_056991 |
| Locus ID | 51056 |
| UniProt ID | P28838 |
| Cytogenetics | 4p15.32 |
| Refseq Size | 2100 |
| Refseq ORF | 1557 |
| Synonyms | HEL-S-106; LAP; LAPEP; PEPS |
| Summary | Presumably involved in the processing and regular turnover of intracellular proteins. Catalyzes the removal of unsubstituted N-terminal amino acids from various peptides.[UniProtKB/Swiss-Prot Function] |
| Protein Families | Druggable Genome, Protease |
| Protein Pathways | Arginine and proline metabolism, Glutathione metabolism, Metabolic pathways |
Documents
| FAQs |
| SDS |
Resources
Recombinant Protein Resources |
Other Versions
{0} Product Review(s)
0 Product Review(s)
Submit review
Be the first one to submit a review
Product Citations
*Delivery time may vary from web posted schedule. Occasional delays may occur due to unforeseen
complexities in the preparation of your product. International customers may expect an additional 1-2 weeks
in shipping.






























































































































































































































































 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China