TDO2 (NM_005651) Human Recombinant Protein
CAT#: TP302777
Recombinant protein of human tryptophan 2,3-dioxygenase (TDO2), 20 µg
View other "TDO2" proteins (3)
Special Offer: Get a 20% discount on this product. Use code: "MVPro20".
Specifications
| Product Data | |
| Species | Human |
| Expression Host | HEK293T |
| Expression cDNA Clone or AA Sequence |
>RC202777 protein sequence
Red=Cloning site Green=Tags(s) MSGCPFLGNNFGYTFKKLPVEGSEEDKSQTGVNRASKGGLIYGNYLHLEKVLNAQELQSETKGNKIHDEH LFIITHQAYELWFKQILWELDSVREIFQNGHVRDERNMLKVVSRMHRVSVILKLLVQQFSILETMTALDF NDFREYLSPASGFQSLQFRLLENKIGVLQNMRVPYNRRHYRDNFKGEENELLLKSEQEKTLLELVEAWLE RTPGLEPHGFNFWGKLEKNITRGLEEEFIRIQAKEESEEKEEQVAEFQKQKEVLLSLFDEKRHEHLLSKG ERRLSYRALQGALMIYFYREEPRFQVPFQLLTSLMDIDSLMTKWRYNHVCMVHRMLGSKAGTGGSSGYHY LRSTVSDRYKVFVDLFNLSTYLIPRHWIPKMNPTIHKFLYTAEYCDSSYFSSDESD TRTRPLEQKLISEEDLAANDILDYKDDDDKV |
| Tag | C-Myc/DDK |
| Predicted MW | 47.7 kDa |
| Concentration | >0.05 µg/µL as determined by microplate BCA method |
| Purity | > 80% as determined by SDS-PAGE and Coomassie blue staining |
| Buffer | 25 mM Tris-HCl, 100 mM glycine, pH 7.3, 10% glycerol |
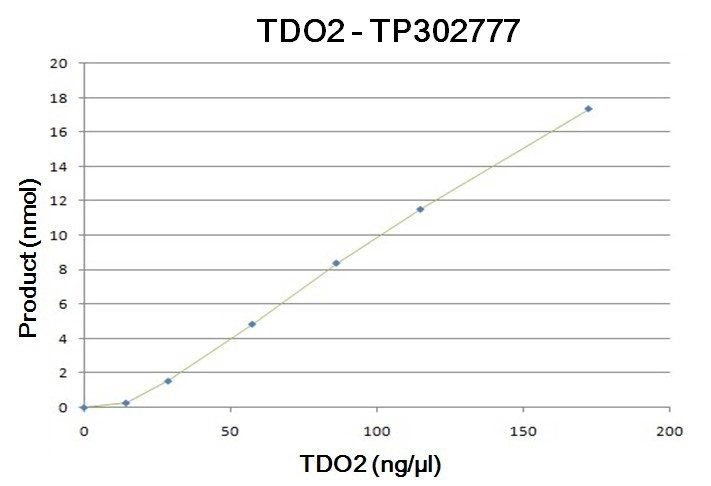
| Bioactivity | The specific activity of TDO2 was determined by monitoring Kynurenine formation from the N-formylkynurenine based on the absorbance at 492nm. The N-formylkynurenine was produced from a conversion of tryptophan with TDO2. The reactions were carried out at 37°C for 40min in 100ul of the reaction volume containing 200mM PBS, pH7.5, 1mM ascorbic acid, and 1.25mM L-tryptophan as the substrate with various amounts of TDO2. The reaction was terminated by adding 50ul of 30% (w/v) trichloroacetic acid. The sample was further incubated for 30min at 60°C and centrifuged at 12000 rpm for 15 min. The supernatant was used to mix with an equal volume of Ehrlich's reagent (2% p-dimethylaminobenza-ldehyde in glacial acetic acid) to measure the absorbance at 492 nm after 10min incubation |
| Preparation | Recombinant protein was captured through anti-DDK affinity column followed by conventional chromatography steps. |
| Note | For testing in cell culture applications, please filter before use. Note that you may experience some loss of protein during the filtration process. |
| Storage | Store at -80°C. |
| Stability | Stable for 12 months from the date of receipt of the product under proper storage and handling conditions. Avoid repeated freeze-thaw cycles. |
| Reference Data | |
| RefSeq | NP_005642 |
| Locus ID | 6999 |
| UniProt ID | P48775 |
| Cytogenetics | 4q32.1 |
| Refseq Size | 1703 |
| Refseq ORF | 1218 |
| Synonyms | HYPTRP; TDO; TO; TPH2; TRPO |
| Summary | This gene encodes a heme enzyme that plays a critical role in tryptophan metabolism by catalyzing the first and rate-limiting step of the kynurenine pathway. Increased activity of the encoded protein and subsequent kynurenine production may also play a role in cancer through the suppression of antitumor immune responses, and single nucleotide polymorphisms in this gene may be associated with autism. [provided by RefSeq, Feb 2012] |
| Protein Pathways | Metabolic pathways, Tryptophan metabolism |
Documents
| FAQs |
| SDS |
Resources
Recombinant Protein Resources |
Other Versions
| SKU | Description | Size | Price |
|---|---|---|---|
| LC401726 | TDO2 HEK293T cell transient overexpression lysate (as WB positive control) |
USD 134.00 |
|
| LY401726 | Transient overexpression lysate of tryptophan 2,3-dioxygenase (TDO2) |
USD 436.00 |
|
| PH302777 | TDO2 MS Standard C13 and N15-labeled recombinant protein (NP_005642) |
USD 3,255.00 |
{0} Product Review(s)
Be the first one to submit a review






























































































































































































































































 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China